What Outcomes Can Be Measured in a Sytematic Review
- Enquiry article
- Open Access
- Published:
Outcome choice and definition in systematic reviews leads to few eligible studies included in meta-analyses: a instance study
BMC Medical Research Methodology book xx, Commodity number:30 (2020) Cite this commodity
Abstract
Background
There is broad recognition of the importance of evidence in informing clinical decisions. When data from all studies included in a systematic review ("review") does not contribute to a meta-analysis, decision-makers can be frustrated. Our objectives were to use the field of eyes and vision every bit a case study and examine the extent to which authors of Cochrane reviews conducted meta-analyses for their review's pre-specified chief outcome domain and the reasons that some otherwise eligible studies were not incorporated into meta-analyses.
Methods
Nosotros examined all completed systematic reviews published by Cochrane Optics and Vision, as of August 11, 2017. We extracted information about each review's outcomes and, using an algorithm, categorized one outcome as its "main" result. Nosotros calculated the percentage of included studies incorporated into meta-analyses for any upshot and for the master outcome. We examined reasons for non-inclusion of studies into the meta-analysis for the main outcome.
Results
We identified 175 completed reviews, of which 125 reviews included ii or more studies. Beyond these 125 reviews, the median proportions of studies incorporated into at least one meta-analysis for whatever outcome and for the chief event were 74% (interquartile range [IQR] 0–100%) and 28% (IQR 0–71%), respectively. Fifty-one reviews (41%) could not conduct a meta-analysis for the main outcome, mostly because fewer than 2 included studies measured the outcome (21/51 reviews) or the specific measurements for the result were inconsistent (sixteen/51 reviews).
Conclusions
Outcome choice during systematic reviews can lead to few eligible studies included in meta-analyses. Core outcome sets and improved reporting of outcomes can assistance solve some of these issues.
Background
At that place is broad recognition of the importance of bear witness in determining clinical controlling [i]. For testify-based healthcare, decision-makers (due east.g., patients, clinicians, guideline developers) increasingly rely on systematic reviews ("reviews") [1]. Reviews identify primary studies, such as clinical trials and observational studies, that have addressed the enquiry question of interest. This research question typically defines the population, interventions, and comparators; these defined aspects in turn assist delineate the chief studies eligible for the review.
Reviews may or may not include quantitative syntheses of data across studies ("meta-analyses"). When accordingly conducted, meta-analyses provide decision-makers with summary estimates (e.g., relative risks) and accompanying estimates of uncertainty (e.g., 95% confidence intervals) that convey information most treatment effectiveness or prophylactic succinctly [2]. Oft, however, meta-analyses cannot exist conducted because the studies address somewhat different clinical questions, assess different outcomes than the systematic reviewer ("reviewer") had pre-specified, are methodologically heterogeneous, or are poorly-reported (e.g., inadequate information about results). In these circumstances, a study may exist eligible for the review, but may not contribute to a meta-analysis [three]. When a review includes multiple studies, simply these studies cannot be included in the meta-analysis, both doers (i.e., reviewers) and users of reviews (i.e., decision-makers) can be frustrated. Decision-makers desire to know how treatments compare quantitatively; they may not exist able to go reliable information about this when only some included studies contribute data to the meta-assay or when no meta-assay is possible [iv].
Outcomes are measures or events used to appraise the effectiveness and/or safety of clinical interventions [5]. A frequent reason for non-acquit of meta-analyses is that the studies assess different outcomes or assess the same outcomes, but do so differently. These scenarios can occur even among high-quality studies.
Although outcomes are fundamental to reviews of interventions, outcomes are typically not considered when determining the eligibility of a primary study in such reviews [6]. This is considering outcomes inform meta-analyses, non whether the main study is eligible for the review. Consistent with guidance in the Cochrane Handbook for Systematic Reviews of Interventions [half-dozen], we believe that studies that accost the population, interventions, and comparators of interest should be included and cataloged in systematic reviews fifty-fifty if they do not report outcomes of interest. Outcome choice in a review is crucial because: (1) outcomes serve as yardsticks for basing conclusions nearly treatments; and (two) which outcomes are chosen and how they are divers can touch on how many meta-analyses can be washed and how many studies can be included in them [7,viii,9,10,xi].
Outcomes may exist assessed differently in different studies because an "outcome" (a seemingly monolithic entity) actually comprises v elements: domain, e.g., visual vigil; specific measurement, e.thousand., Snellen nautical chart; specific metric, eastward.g., ≥3 lines of vision lost; method of aggregation, e.g., proportion; and fourth dimension-points, e.k., 6 months [9, 12]. Some other instance of the application of this v-element framework to clearly specify a particular information point of involvement related to the outcome of "feet" is mean (method of assemblage) change (specific metric) in anxiety (domain) measured through the Hamilton Anxiety Rating Scale (specific measurement) from baseline to 1 twelvemonth (fourth dimension-point) [9, 12].
Nosotros previously demonstrated, through case studies in the fields of optics and vision [11] and HIV/AIDS [10], that reviewers and clinical trialists addressing the same research question oft examine dissimilar outcomes. In improver, inconsistency in event reporting across eligible studies prevents incorporation of all eligible studies into meta-analyses. For instance, a 2022 Cochrane systematic review comparing non-steroidal anti-inflammatory drugs (NSAIDs) with corticosteroids for inflammation after cataract surgery [xiii] included 48 trials, none of which reported data for the review's pre-specified primary effect, "proportion of patients with intraocular inflammation at 1 week after surgery."
To document the extent and determinants of this problem, we embarked on the current case report in the field of eyes and vision. Our objectives were to examine the extent to which Cochrane reviews in eyes and vision conducted meta-analyses for the master outcome domain and the reasons why some otherwise eligible studies were not incorporated into meta-analyses.
Methods
Reviews examined
We examined all completed systematic reviews published by Cochrane Eyes and Vision in the Cochrane Database of Systematic Reviews as of Baronial 11, 2017. We excluded reviews that were still in the protocol stage.
Data extraction
We adult a data extraction form in the Systematic Review Data Repository (SRDR), an open-source platform for extracting and archiving information [14, xv]. Using a airplane pilot-tested form, two individuals (from among SM, HK, BTS, and IJS) independently extracted data, resolving discrepancies through discussion. We extracted the post-obit data: yr published, population (i.e., eye office/region affected), and types of interventions and comparators. We extracted the numbers of master, secondary, and other, i.e., non-primary and not-secondary, outcome domains. We also extracted the number of studies included in the review and in ≥1 meta-analysis for any, any primary, any secondary, and whatsoever other domain.
"Main" outcome domains
We categorized one domain from each review equally its "main" upshot domain (Table 1). For reviews that named only one master outcome domain, we categorized it as the chief outcome domain; for reviews that named more than than one principal outcome domain (or named more than 1 secondary consequence domain), we categorized the primary event domain (or secondary event domain) with the highest number of included studies every bit the main issue domain. For reviews that did non name whatever primary or secondary outcome domains, nosotros categorized the "other", i.e., nonprimary and non-secondary, consequence domain with the highest number of included studies every bit the main result domain.
For each main outcome domain, we extracted the other four elements specified: specific measurement, specific metric, method of aggregation, and fourth dimension-points. For the primary outcome domain, we also extracted the numbers of studies that reported measuring information technology, reported any data, reported whatever meta-analyzable data, and were incorporated into ≥ 1 meta-analysis. We considered data for a given outcome from a given written report to be "meta-analyzable" if the written report reported acceptable data then that it could be incorporated into a meta-analysis. For chiselled outcomes, meta-analyzable meant that either of these conditions were met: (1) total number of participants and number of participants with the outcome were reported for each study arm; and (2) the betwixt-grouping handling effect (e.chiliad., relative risk) and an uncertainty guess (e.g., 95% confidence interval) were reported. For continuous and time-to-event outcomes, meta-analyzable meant that either of these conditions were met: (1) hateful and uncertainty estimates were reported for each study arm; and (2) the between-group treatment effect (e.grand., mean divergence) and an uncertainty estimate were reported.
Results
Reviews examined
We identified 175 completed systematic reviews published by Cochrane Eyes and Vision in the Cochrane Database of Systematic Reviews (Tabular array two). The reviews were published between Jan 1, 2005 and August 11, 2022 (median = 2014). The virtually common populations were patients with retinal/choroidal disease (35 reviews; 20%) and visual harm/low vision (33 reviews; 19%). The almost common types of interventions/comparators were drugs (74 reviews; 42%) and surgeries (67 reviews; 38%).
Incorporation of studies into meta-analyses for any consequence domain
The 175 included reviews examined a median of 6 total outcome domains, including a median of 1 primary outcome domain, 4 secondary event domains, and i other outcome domain.
The 175 reviews included a median of 3 studies (IQR 1–nine); 125 reviews (71%) included ≥2 studies. For these 125 reviews, Fig. 2 plots the percent of studies incorporated into a meta-assay for whatsoever consequence domain (blueish line) and for the main outcome domain (red bars). Amid these reviews, 44/125 reviews (35%) incorporated every included study into ≥ane meta-assay (for any outcome domain). Conversely, 33/125 reviews (26%) did not incorporate any study into any meta-assay for any outcome, i.eastward., they did non carry any meta-analysis. The remaining 48/125 reviews (38%) incorporated only a subset of their studies into ≥ane meta-analysis. These 48 reviews included a median of 12.5 studies (IQR 6–22), and the meta-analyses in these reviews incorporated a median of 6.5 studies (IQR 4–13).
Among the 125 reviews that could take conducted a meta-assay, i.eastward., those including ≥2 studies, the median proportion of studies incorporated into ≥1 meta-analysis for whatsoever upshot was 74% (IQR 0–100%). Amongst the 92 reviews that conducted a meta-assay, the median proportion of studies incorporated into ≥1 meta-assay for any outcome was 93% (IQR 64–100%).
Characteristics of master outcome domains
Almost all reviews (172/175 reviews; 98%) named ≥1 primary outcome domain (Table 1). Three in four reviews (131/175 reviews; 75%) each named exactly one principal event domain, which nosotros categorized every bit their main outcome domain. The near frequent main result domains across the 175 reviews were visual acuity (31%) and intraocular force per unit area (half-dozen%) (Table iii). Thirty-8 outcome domains were main outcome domains in just one review each. The main consequence was categorical in 70% and continuous in 29% of reviews. Most main outcome domains (98%) were efficacy outcomes, i.due east., non safety outcomes.
Incorporation of studies into meta-analyses for the main outcome domain
Amid the 125 reviews including ≥two studies, simply 18 reviews (xiv%) incorporated all their studies into a meta-analysis for the main outcome domain. Conversely, 51/125 reviews (41%) did not incorporate any study into the meta-analysis for the master consequence domain, i.e., they did not conduct any meta-analysis for the chief event domain. The remaining 56/125 reviews (45%) incorporated but a subset of their studies into the meta-analysis for the chief event domain. These 56 reviews included a median of 12 studies each, and the meta-analyses for the main outcome domain in these reviews incorporated a median of iv studies each.
Among the 125 reviews that could have conducted a meta-analysis, i.e., those including ≥2 studies, the median proportion of studies incorporated into ≥1 meta-analysis for the principal outcome domain was 28% (IQR 0–71%). Among the 74 reviews that conducted meta-analyses for the main outcome domain, the median proportion of studies incorporated was 67% (IQR 39–91%).
Meta-analysis acquit for the chief outcome domain
Effigy 1 illustrates a cascading effect of loss of information every bit regards the main outcome domain in the 175 reviews. Thirty-five reviews (twenty%) included no studies, i.eastward., were empty reviews, and 15 (9%) included ane written report each (Fig. 1). Of the 125 reviews including ≥two studies, i.east., those in which a meta-analysis could theoretically be done for the master outcome if ≥2 studies reported meta-analyzable data, only 74 reviews (59%) conducted a meta-analysis for the main outcome.
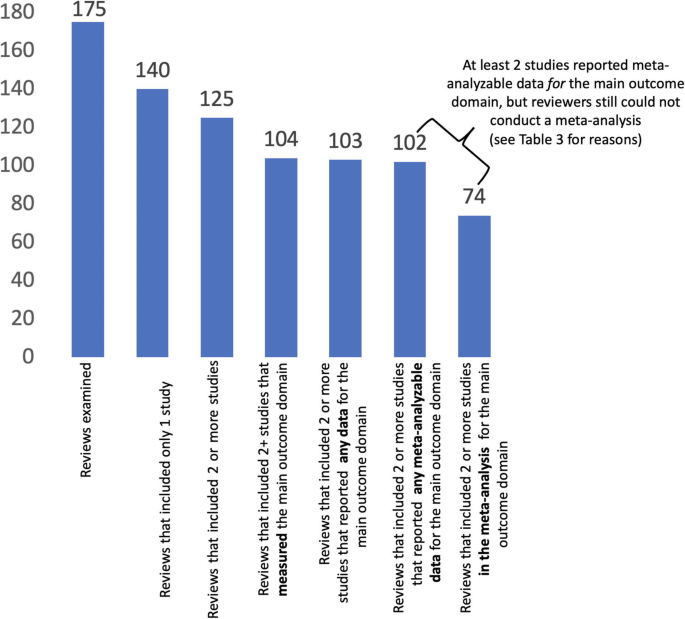
Deport of meta-analyses for the primary effect domain
Reasons for non-conduct of meta-analyses for the main outcome domain
Amidst the 125 reviews including ≥2 studies, 51 reviews (41%) did not conduct a meta-analysis for the main outcome domain. For 21/51 reviews (41%), fewer than two studies measured the review's main outcome (Table 4). When ≥2 studies reported meta-analyzable information, at that place were numerous reasons why reviewers did not conduct a meta-assay, most oft due to inconsistency in issue elements amid the included studies. Specifically, data could not be meta-analyzed because the specific measurements used (sixteen/51 reviews; 31%) and time-points examined (9/51 reviews; 18%) were inconsistent among studies.
Effigy 2 demonstrates that the loss of information for the main issue domain (scarlet confined) was similar in design to the loss of information when considering any outcome domain (bluish line).
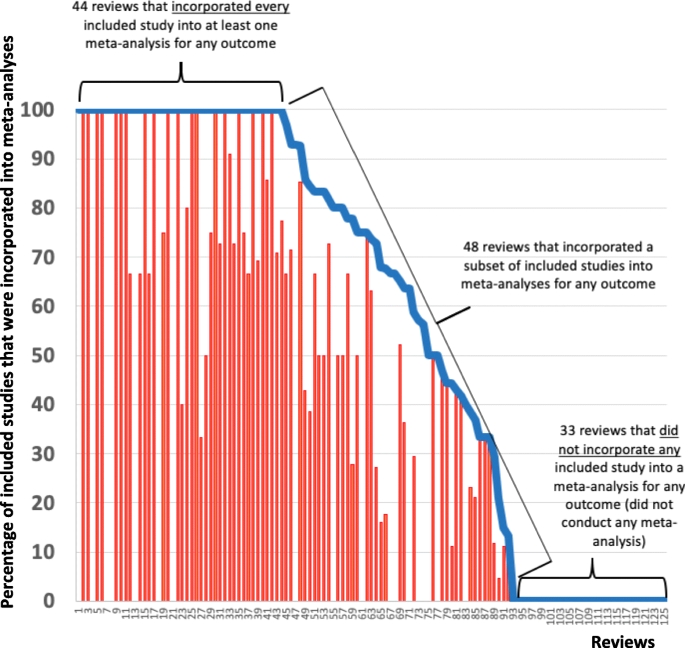
Percent of studies included in the review that were incorporated into a meta-analysis for any event (blueish line) and for the review'due south main effect (red bars)
Notes: This Effigy excludes the 50 systematic reviews in whom a meta-analysis was not possible: 35 systematic reviews that each included 0 studies (i.east., "empty reviews") and 15 systematic reviews included that each included only 1 written report. When the blue line is non-0 simply the reddish bars are 0, information technology implies that the systematic review did not deport a meta-assay for the main result, simply did so for ≥1 of the remaining outcomes
Give-and-take
Through a example study of all Cochrane reviews in the field of eyes and vision, the current work demonstrates three major areas that need comeback.
First, primary studies addressing similar enquiry questions should marshal their outcomes better. Studies frequently could not be incorporated into meta-analyses because the outcomes were not aligned, either because the domains or ≥1 of the other four outcome elements did non overlap. Amidst the reviews including ≥2 studies, only 59 and 74% could bear a meta-analysis for the primary outcome and for any result, respectively. In other words, even when reviews included ≥2 studies, 41 and 26% of reviews missed opportunities to bear a meta-analysis to succinctly convey data regarding the main outcome and whatsoever upshot, respectively.
2d, reviews and main studies should marshal their outcomes better. When looking at reviews that could accept conducted a meta-analysis, i.e., those including ≥two studies, the median percentages of included studies incorporated into the meta-analysis for the master outcome and for any outcome were 28 and 74%, respectively. This suggests that, approximately 7 in 10 studies that reviewers include are not incorporated into the meta-analysis for the principal outcome, and 1 in 4 studies are non incorporated into the meta-analysis for any outcome. In previous piece of work, nosotros demonstrated poor overlap between outcomes in clinical trials and reviews, and possible differences in the types of outcomes they examine [10, 11]. For HIV/AIDS, we demonstrated that reviewers examined more long-term clinical outcomes and patient-centered outcomes than did clinical trialists. Such differences may arise considering: (i) reviews may more than directly inform clinical practice guidelines, and (2) reviewers may be less affected by common constraints faced by clinical trialists, east.g., costs and sample size [10].
Our findings beg the question of who should prioritize outcomes for measurement and reporting in research. It has aptly been stated that achieving consensus in outcome use across research "cannot exist left to serendipity." [xvi] One deliberate and cardinal aspect of the solution to the problem of result inconsistency is the development of "core event sets." A core outcome ready is a minimum set of outcomes that should be measured and reported in all clinical trials addressing a given condition [17]. Cadre consequence sets are increasingly mutual in diverse health fields; a 2022 systematic review identified 307 cadre outcomes sets [18]. However, outcome inconsistency remains widespread; 40% of recent (2019) published Cochrane reviews explicitly noted this problem [19].
We [ten, xi] and others [20] accept argued that, every bit stakeholders in a given field, systematic reviewers should both participate in the development of and adopt cadre outcome sets for that field. By broadening the participation in outcome prioritization efforts, this could potentially help ensure that the outcomes that are measured and reported in research are widely relevant and important. Two aspects of core outcome sets are worthy of description. First, core outcome sets practice not stifle innovation; they are but meant to stand for a minimum set up of outcomes that should be reported. One time a core outcome set exists for a given topic, clinical trialists working in that topic area should explicitly specify the intention to measure and report the outcomes in the set. Second, cadre effect sets are not static; they can and should be updated as the field advances and new cognition emerges.
The 3rd major area in need of improvement that our written report demonstrates is the reporting of outcomes in primary studies. Results data from principal studies were often not meta-analyzable even when outcomes might have been aligned. In add-on, outcome domains were frequently not reported in primary studies or ≥1 of the event elements were frequently missing or inadequately reported (e.g., "worsening of disease" without clarification of how "worsening" was defined). It is possible that the studies measured these outcomes, but did not report measuring them or reported them inadequately. If such selective reporting, either non-reporting or inadequate reporting, of outcomes in the included studies occurred every bit a function of the management of the outcome'southward results, it would exist suggestive of outcome reporting bias [21]. In this case study, we relied on the reviewers' reporting of the extent to which the principal studies reported the outcomes. Because nosotros did not examine the reports of the chief studies (or their protocols), nosotros are unable to comment definitively on whether not-reporting of the outcomes indicates result reporting bias. Withal, outcome reporting bias in primary studies has been documented to be a widespread trouble across reviews [22,23,24,25,26], and, every bit such, is a likely explanation for some outcomes not being reported.
Implications
For the evidence-based medicine paradigm to work, decision-makers must exist able to rely on systematic reviews, which in turn rely on the results of primary studies. For results of primary studies to be actionable, there (1) needs to be alignment in outcomes considered important to both primary study researchers and reviewers, and (2) those outcomes need to be reported completely. Important discussions need to be had regarding who should choose outcomes for the field and how such choices should exist made. We, in conjunction with others, suggest that these discussions should include, at the to the lowest degree, clinicians, patients, clinical trialists, systematic reviewers, regulators, and other determination-makers [27].
We have demonstrated that the option of outcomes for systematic reviews may have led to loss of information through non-incorporation of results from included studies into meta-analyses. The well-nigh substantial drops in the percentage of reviews conducting meta-analyses for the main outcome domain appeared to be due to inadequate numbers of studies reporting the outcome and, when at that place were adequate numbers of studies for a meta-analysis (i.e., ≥2 studies), differences in the specific measurements and time-points used.
Our findings also demonstrate that fifty-fifty when focusing on reviews that conducted meta-analyses for their main outcome domain, only nigh 2 in three studies were incorporated into those meta-analyses. As such, non-incorporation of included studies into meta-analyses represents two main problems. First, it represents missed opportunities for using research to inform determination-making through testify synthesis. This contributes considerably towards research waste [28,29,xxx]. 2nd, non-incorporation of included studies into meta-analyses represents a failed obligation on the function of the researchers (both trialists and reviewers) [31]. As a community of researchers, both parties have a solemn obligation to enquiry participants to ensure that their participation will lead to a useful contribution to scientific discipline; declining to hold upon outcomes that should be collected and adequately reported likely violates this obligation.
Other solutions
Core outcome sets are integral to solving the problems this written report illustrates. Other parts of the solution are worth discussing. We agree with existing recommendations against studies existence excluded from systematic reviews solely on the basis of the lack of relevant outcome data [three]. Thankfully, such recommendations have been associated with a reduction in the number of reviews excluding studies solely on the basis of outcome data [32]. Every bit the current written report demonstrates, the review team's pick of outcomes may not align with that of the primary studies. This may exist particularly true for eyes and vision, a field with few core outcome sets [iv, 18]. We also encourage reviewers to report an outcome matrix [23, 24], a transparent and simple manner to bespeak all fully-reported, partially-reported, or non-reported outcomes in each included study.
Large numbers of empty reviews and reviews including only i written report
Twenty-percent of the reviews we examined were empty and 9% included but one study each. While such reviews are useful in driving primary research, the possible reasons for the paucity of studies in them are worth exploring. One possibility is that these represent topics that primary researchers have not still studied. Another is that but observational studies addressing these topics may exist; Cochrane reviews typically include simply randomized trials. It as well is possible that these topics reverberate the priorities of Cochrane Eyes and Vision and the authors of these reviews, rather than of the field at-large.
Limitations
Our study has sure limitations. Showtime, we focused on Cochrane reviews within i field. Loss of information due to the choice of review outcomes could exist a bigger, similar, or smaller problem in not-Cochrane systematic reviews in eyes and vision or systematic reviews in other fields. Second, we analyzed in-depth the extent of incorporation of included studies into meta-analyses only for the primary event domain. Meta-analyses of other principal, secondary, and other outcome domains may have incorporated college percentages of included studies. However, Fig. 2 suggests that this is probable not the instance. Information technology is possible that our algorithm for categorizing the "main" issue for each review could accept impacted our findings. Just, in reviews where more than 1 outcome domain could accept served as the main issue, we categorized as the main effect the outcome that the highest number of included studies had reported. Our results thus represent the best-case scenario. Third, nearly outcome domains (98%) were efficacy outcomes. Selective result reporting has also been reported to be a trouble for prophylactic outcomes [33]. 4th, we relied on the reviews to determine whether or not each included study did the following for the main outcome domain: reported measuring it, reported whatsoever results for information technology, and reported meta-analyzable data for it. Related to this, we did non examine the ceremoniousness or feasibility of the reviewers' being able to deport meta-analyses when the included studies reported data in a format different from what the reviewers were interested. As such, our results certificate what was actually washed in the reviews.
Conclusions
This case study of all Cochrane systematic reviews addressing an unabridged field (eyes and vision) demonstrates that only 59 and 74% of the reviews including ≥ii studies could comport a meta-assay for the principal outcome and for any effect, respectively. In evidence-based healthcare, such loss of information represents missed opportunities and a failed obligation by researchers to research participants to ensure that their participation will lead to a useful contribution to science. Cadre outcome sets and improved event reporting can aid solve some of these problems.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable asking.
Abbreviations
- IQR:
-
Interquartile range
- NSAID:
-
Non-steroidal anti-inflammatory drug
- SRDR:
-
Systematic Review Data Repository
References
-
Institute of Medicine. Finding what works in health care: standards for systematic reviews. Washington, DC: The National Academies Press; 2011.
-
Borenstein M, Hedges LV, Higgins JP, Rothstein HR. A basic introduction to fixed-outcome and random-effects models for meta-assay. Res Synth Methods. 2010;i(ii):97–111.
-
Higgins JPT, Lasserson T, Chandler J, Tovey D, Churchill R. Methodological Expectations of Cochrane Intervention Reviews. Version one.02 ed. London: Cochrane; 2016.
-
Saldanha IJ, Le JT, Solomon SD, Repka MX, Akpek EK, Li T. Choosing Core outcomes for utilize in clinical trials in ophthalmology: perspectives from three ophthalmology outcomes working groups. Ophthalmology. 2019;126(1):six–9.
-
Meinert CL. Clinical trials dictionary: terminology and usage recommendation. 2nd ed Hoboken, NJ: Wiley; 2012.
-
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors). Cochrane Handbook for Systematic Reviews of Interventions. 2nd Edition. Chichester: Wiley; 2019.
-
Mayo-Wilson Due east, Li T, Fusco N, et al. Cherry-picking past trialists and meta-analysts tin bulldoze conclusions well-nigh intervention efficacy. J Clin Epidemiol. 2017;91:95–110.
-
Mayo-Wilson East, Li T, Fusco N, Dickersin K. Practical guidance for using multiple data sources in systematic reviews and meta-analyses (with examples from the MUDS study). Res Synth Methods. 2018;ix(ane):2–12.
-
Saldanha IJ, Dickersin G, Wang Ten, Li T. Outcomes in Cochrane systematic reviews addressing 4 common center atmospheric condition: an evaluation of completeness and comparability. PLoS One. 2014;9(ten):e109400.
-
Saldanha IJ, Li T, Yang C, Owczarzak J, Williamson PR, Dickersin M. Clinical trials and systematic reviews addressing similar interventions for the same condition practice non consider similar outcomes to be important: a example study in HIV/AIDS. J Clin Epidemiol. 2017;84:85–94.
-
Saldanha IJ, Lindsley K, Do DV, et al. Comparing of clinical trial and systematic review outcomes for the 4 Most prevalent middle diseases. JAMA Ophthalmology. 2017;135(9):933–40.
-
Zarin DA, Tse T, Williams RJ, Califf RM, ide NC. The ClinicalTrials.gov results database--update and key issues. Due north Engl J Med. 2011;364(9):852–60.
-
Juthani VV, Clearfield E, Chuck RS. Non-steroidal anti-inflammatory drugs versus corticosteroids for controlling inflammation after uncomplicated cataract surgery. Cochrane Database Syst Rev. 2017;seven:Cd010516.
-
Ip Due south, Hadar N, Keefe Southward, et al. A web-based annal of systematic review data. Syst Rev. 2012;1:xv.
-
Li T, Vedula SS, Hadar North, Parkin C, Lau J, Dickersin K. Innovations in data collection, direction, and archiving for systematic reviews. Ann Intern Med. 2015;162(4):287–94.
-
Clarke Grand. Standardising outcomes for clinical trials and systematic reviews. Trials. 2007;8:39.
-
Williamson PR, Altman DG, Blazeby JM, et al. Developing cadre effect sets for clinical trials: issues to consider. Trials. 2012;13:132.
-
Gargon E, Gorst SL, Harman NL, Smith V, Matvienko-Sikar K, Williamson PR. Choosing important health outcomes for comparative effectiveness enquiry: 4th annual update to a systematic review of core outcome sets for research. PLoS One. 2018;xiii(12):e0209869.
-
Williamson PR, Oliveira RD, Clarke K, et al. The relevance and uptake of core outcome sets in Cochrane systematic reviews Submitted.
-
Clarke Thou, Williamson PR. Core issue sets and systematic reviews. Syst Rev. 2016;5:11.
-
Chan AW, Krleza-Jeric K, Schmid I, Altman DG. Outcome reporting bias in randomized trials funded by the Canadian Institutes of Health Research. CMAJ. 2004;171(seven):735–40.
-
Dwan K, Kirkham JJ, Williamson PR, Hazard C. Selective reporting of outcomes in randomised controlled trials in systematic reviews of cystic fibrosis. BMJ Open. 2013;3(6).
-
Kirkham JJ, Altman DG, Chan AW, Gamble C, Dwan KM, Williamson PR. Issue reporting bias in trials: a methodological approach for assessment and adjustment in systematic reviews. BMJ. 2018;362:k3802.
-
Kirkham JJ, Dwan KM, Altman DG, et al. The impact of outcome reporting bias in randomised controlled trials on a cohort of systematic reviews. BMJ. 2010;340:c365.
-
Kirkham JJ, Riley RD, Williamson PR. A multivariate meta-assay approach for reducing the touch of outcome reporting bias in systematic reviews. Stat Med. 2012;31(twenty):2179–95.
-
Page MJ, McKenzie JE, Kirkham J, et al. Bias due to selective inclusion and reporting of outcomes and analyses in systematic reviews of randomised trials of healthcare interventions. Cochrane Database Syst Rev. 2014;(10):Mr000035.
-
Williamson PR, Altman DG, Bagley H, et al. The COMET Handbook: version i.0. Trials. 2017;xviii(Suppl iii):280.
-
Chalmers I, Bracken MB, Djulbegovic B, et al. How to increase value and reduce waste when research priorities are set. Lancet (London, England). 2014;383(9912):156–65.
-
Clarke K, Brice A, Chalmers I. Accumulating research: a systematic account of how cumulative meta-analyses would have provided knowledge, improved health, reduced damage and saved resources. PLoS One. 2014;9(vii):e102670.
-
Macleod MR, Michie S, Roberts I, et al. Biomedical research: increasing value, reducing waste product. Lancet (London, England). 2014;383(9912):101–4.
-
Law A, Lindsley Grand, Rouse B, Wormald R, Dickersin K, Li T. Missed opportunity from randomised controlled trials of medical interventions for open-bending glaucoma. Br J Ophthalmol. 2017;101(10):1315–7.
-
Dwan KM, Williamson PR, Kirkham JJ. Do systematic reviews still exclude studies with "no relevant effect data"? BMJ. 2017;358:j3919.
-
Saini P, Loke YK, Take chances C, Altman DG, Williamson PR, Kirkham JJ. Selective reporting bias of harm outcomes within studies: findings from a cohort of systematic reviews. BMJ. 2014;349:g6501.
Acknowledgements
The authors admit Elizabeth Clearfield and Dr. Roy Chuck for their contributions to the concepts presented in this work.
Funding
The National Institutes of Health (Grant Number UG EY020522) funded this work. The funder played no role in the design of the report; collection, analysis, and interpretation of the data, or writing of the manuscript.
Writer information
Affiliations
Contributions
IJS, KL, and KD conceptualized and designed the study. IJS and KD analyzed and interpreted the data. IJS drafted the manuscript. All authors provided disquisitional feedback to the manuscript. All authors read and canonical the final manuscript.
Corresponding author
Ethics declarations
Ethics blessing and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Drs. Saldanha and Dickersin and Ms. Lindsley and Money declare affiliation with Cochrane Eyes and Vision during conduct of the work related to this manuscript. Ms. Kimmel and Mr. Smith were research assistants on this study. All of the reviews examined in this manuscript were produced by Cochrane Eyes and Vision. No other disclosures are reported.
Boosted data
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open up Access This commodity is distributed nether the terms of the Creative Commons Attribution iv.0 International License (http://creativecommons.org/licenses/past/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Eatables Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/ane.0/) applies to the data made available in this commodity, unless otherwise stated.
Reprints and Permissions
About this article
Cite this article
Saldanha, I.J., Lindsley, K.B., Money, S. et al. Upshot choice and definition in systematic reviews leads to few eligible studies included in meta-analyses: a case study. BMC Med Res Methodol 20, thirty (2020). https://doi.org/10.1186/s12874-020-0898-2
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s12874-020-0898-2
Keywords
- Systematic review
- Outcomes
- Meta-analysis
- Core consequence sets
- Loss of data
- Clinical trials
Source: https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-020-0898-2#:~:text=Outcomes%20are%20measures%20or%20events,outcomes%2C%20but%20do%20so%20differently.
0 Response to "What Outcomes Can Be Measured in a Sytematic Review"
Post a Comment